These symbols will represent the atoms present in the covalent bond. Method 1 of Write the atomic symbol for each atom. Bdo evasion gearĮxplore this Article methods. By using our site, you agree to our cookie policy.
#How to do a lewis structure for ch4 on chemdoodle for free
Please help us continue to provide you with our trusted how-to guides and videos for free by whitelisting wikiHow on your ad blocker. For more tips from our Science co-author, including how to make Lewis structures for larger covalent molecules or ions, read on!ĭid this summary help you? Yes No. Finally, represent any remaining unbonded electrons by drawing dots around their respective atoms. Next, draw lines between the atoms to represent that bond.įor example, use 1 line to show a single bond, or draw 2 lines if they have a double bond. Then, determine whether the atoms are held together by a single, double, or triple bond. To draw Lewis dot structures, start by writing the atomic symbols for the 2 atoms side-by-side. The complexity of the drawing will vary depending on whether you are creating a Lewis dot structure for a diatomic 2-atom covalent molecule, a larger covalent molecule, or ionically bonded molecules. However, these structures are helpful in understanding the bonding and valence electron configurations of different atoms and molecules.
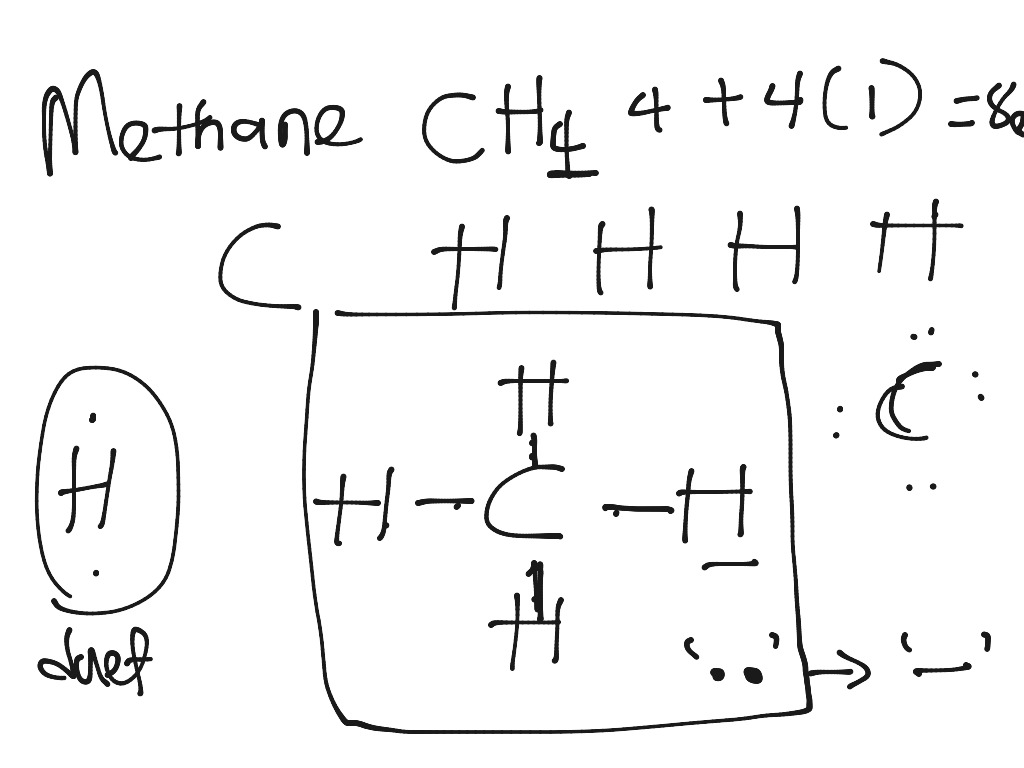
Drawing Lewis dot structures also known as Lewis structures or Lewis diagrams can be confusing, particularly for a beginning chemistry student. There are 13 references cited in this article, which can be found at the bottom of the page. Her studies are focused on proteins and neurodegenerative diseases. This article was co-authored by Meredith Juncker, PhD.
